MFDA Warns Against Unauthorised Sale of Semaglutide and Azelaic Acid
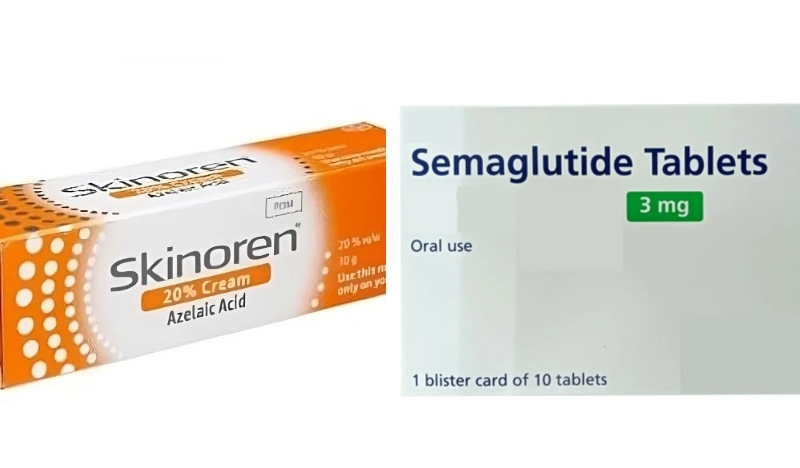
According to MedlinePlus, Semaglutide is used in the treatment of diabetes to help control blood sugar levels, while Azelaic Acid is prescribed to treat skin conditions such as rosacea.
The Maldives Food and Drug Authority (MFDA) has warned against the unauthorised sale and marketing of prescription Semaglutide and Azelaic Acid on social media.
In a statement, the MFDA said it has recently observed social media pages engaging in the unauthorised sale of medicines that should only be used under medical supervision and with a valid prescription. The authority said these products are being promoted as beauty or cosmetic items, despite their classification as prescription-only medicines.
The MFDA specifically identified Semaglutide and Azelaic Acid as among the medicines being marketed and sold online without authorisation. According to MedlinePlus, a US government-run website which is part of the National Institute of Health (NIH), Semaglutide is used in the treatment of diabetes to help control blood sugar levels, while Azelaic Acid is prescribed to treat skin conditions such as rosacea. The authority stressed that both medicines must only be used under the guidance of a qualified medical professional.
pic.twitter.com/t3fA1KcDCN— MFDA (@MFDA_mv) January 19, 2026
The authority warned that the unsupervised and improper use of such medicines could lead to serious health risks and long-term consequences.
MFDA said any individual or business found to have imported, sold or marketed medicines without obtaining the required permits and approvals from relevant authorities will face regulatory action.
Yesterday, the MDFA urged the public to ensure they purchase medicines only from pharmacies that are officially registered and authorised by the authority. The authority stressed that medicines can only be imported and sold with its approval, and that pharmacies must hold a valid licence. It also noted that pharmacy staff are required to obtain formal authorisation, and that working without a valid pharmacist identification card is prohibited.
The authority advised the public to check that pharmacies are licensed, pharmacists carry valid ID cards, medicines are properly packaged, expiry dates are clearly marked, and usage instructions are provided. The reminder comes amid ongoing public concerns over medicine shortages and the circulation of substandard imported drugs, issues authorities say they are continuing to address.